Balloon Pulmonary Angioplasty (BPA)
Percutaneous BPA is an emerging interventional “keyhole” technique for patients with inoperable CTEPH. Occluded pulmonary vessels are dilated through the insertion and inflation of a small balloon.1
Balloon pulmonary angioplasty.2
BPA should only be performed in an expert center in the management of CTEPH.2
To reduce the risk of complications and achieve therapeutic benefit, this procedure is generally:2,3
* repeated several times (4–6 sessions per patient, at 2–8-week intervals) and
* performed in multiple vessel locations during each session.
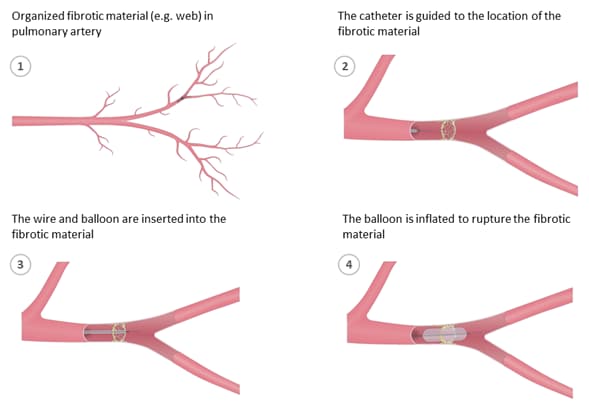
BPA benefits2–4
* Improvement in pulmonary hemodynamics, symptoms, and functional capacity seen in eight observational studies (considered moderate-quality evidence) of BPA.
BPA risks2–4
* Complications include mortality resulting from the procedure, perforation, aneurysm, reperfusion pulmonary edema, hemoptysis/hemosputum, wire injury, vessel dissection, vessel rupture, pulmonary parenchymal bleeding and hemorrhagic pleural effusions.
* Acute mortality (<30 days post-BPA) ranges from 1.4% to 10%.
Use of targeted medical therapy alongside BPA is common practice and can stabilize patient hemodynamics and improve right heart function.
The RACE (Riociguat versus balloon pulmonary Angioplasty in non-operable Chronic thromboEmbolic pulmonary hypertension) trial (NCT 02634203) is evaluating the safety and efficacy of riociguat versus BPA in patients with non-operable CTEPH.5
You may also be interested in
References:
1. Gall H et al. Eur Respir Rev 2017;26:160121. 2.Lang I et al. Eur Respir Rev 2017;26:160119. 3.National Health Service (NHS) England. Clinical Commissioning Policy: Balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension (all ages). Available at: https://www.england.nhs.uk/wp-content/uploads/2018/04/ccp-balloon-pulmonary-angioplasty-for-ctph.pdf (accessed August 2018). 4.Phan K et al. Heart Lung Circ 2018;27:89–98. 5.US National Library of Medicine. ClincialTrials.gov. Riociguat Versus Balloon Pulmonary Angioplasty in Non-operable Chronic thromboEmbolic Pulmonary Hypertension (RACE). NCT02634203. Available at: https://clinicaltrials.gov/ct2/show/NCT02634203 (accessed August 2018).


